What is the source of energy that changes water from liquid to gas
3.2: Energy of Phase Changes
- Folio ID
- 164741
Learning Objectives
- To summate the energy changes that accompany phase changes.
We accept advantage of changes between the gas, liquid, and solid states to cool a drink with ice cubes (solid to liquid), cool our bodies by perspiration (liquid to gas), and absurd nutrient within a refrigerator (gas to liquid and vice versa). We use dry ice, which is solid CO2, as a refrigerant (solid to gas), and we brand bogus snow for skiing and snowboarding by transforming a liquid to a solid. In this section, we examine what happens when any of the three forms of affair is converted to either of the other 2. These changes of state are frequently called stage changes. The 6 most mutual stage changes are shown in Figure \(\PageIndex{ane}\).
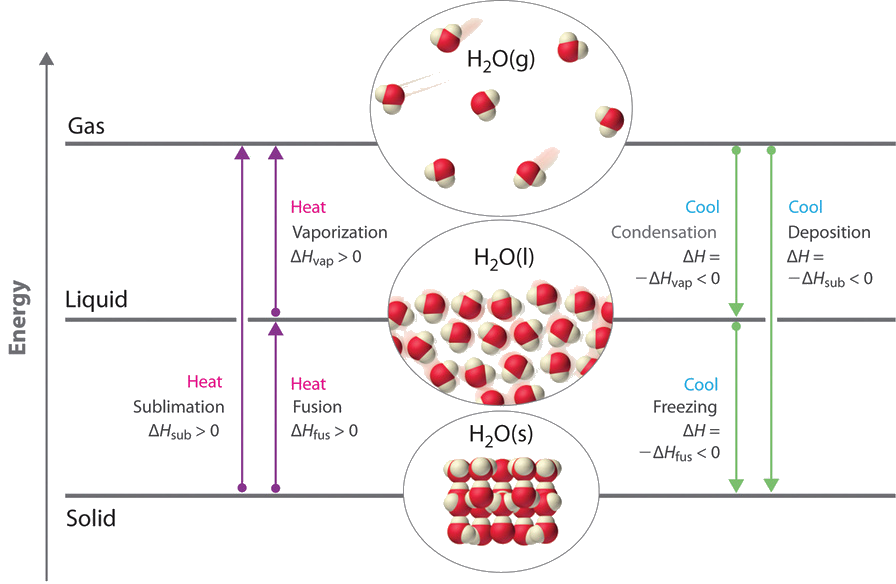
Energy Changes That Accompany Phase Changes
Stage changes are always accompanied by a change in the energy of a arrangement. For instance, converting a liquid, in which the molecules are close together, to a gas, in which the molecules are, on average, far apart, requires an input of free energy (estrus) to give the molecules plenty kinetic energy to allow them to overcome the intermolecular attractive forces. The stronger the attractive forces, the more energy is needed to overcome them. Solids, which are highly ordered, have the strongest intermolecular interactions, whereas gases, which are very matted, have the weakest. Thus any transition from a more ordered to a less ordered country (solid to liquid, liquid to gas, or solid to gas) requires an input of energy; information technology is endothermic. Conversely, any transition from a less ordered to a more than ordered country (liquid to solid, gas to liquid, or gas to solid) releases energy; it is exothermic. The energy change associated with each common stage change is shown in Figure \(\PageIndex{one}\).
ΔH is positive for whatever transition from a more ordered to a less ordered country and negative for a transition from a less ordered to a more than ordered state.
Previously, we defined the enthalpy changes associated with various chemical and physical processes. The melting points and molar enthalpies of fusion (\(ΔH_{fus}\)), the energy required to convert from a solid to a liquid, a process known as fusion (or melting), as well as the normal boiling points and enthalpies of vaporization (\(ΔH_{vap}\)) of selected compounds are listed in Table \(\PageIndex{i}\).
| Substance | Melting Point (°C) | ΔHfus (kJ/mol) | Boiling Point (°C) | ΔHvap (kJ/mol) |
|---|---|---|---|---|
| N2 | −210.0 | 0.71 | −195.eight | 5.half dozen |
| HCl | −114.ii | 2.00 | −85.1 | 16.2 |
| Br2 | −7.two | 10.half dozen | 58.8 | 30.0 |
| CCl4 | −22.6 | two.56 | 76.eight | 29.8 |
| CH3CH2OH (ethanol) | −114.one | 4.93 | 78.iii | 38.6 |
| CH3(CH2)4CH3 (n-hexane) | −95.four | 13.1 | 68.7 | 28.9 |
| HiiO | 0 | vi.01 | 100 | forty.seven |
| Na | 97.8 | 2.6 | 883 | 97.four |
| NaF | 996 | 33.4 | 1704 | 176.i |
The substances with the highest melting points usually accept the highest enthalpies of fusion; they tend to be ionic compounds that are held together by very strong electrostatic interactions. Substances with loftier boiling points are those with strong intermolecular interactions that must be overcome to convert a liquid to a gas, resulting in high enthalpies of vaporization. The enthalpy of vaporization of a given substance is much greater than its enthalpy of fusion because information technology takes more energy to completely separate molecules (conversion from a liquid to a gas) than to enable them only to motion past one another freely (conversion from a solid to a liquid).
Less energy is needed to let molecules to move by each other than to separate them totally.
The straight conversion of a solid to a gas, without an intervening liquid phase, is chosen sublimation. The amount of energy required to sublime 1 mol of a pure solid is the enthalpy of sublimation (ΔH sub). Mutual substances that sublime at standard temperature and pressure (STP; 0°C, 1 atm) include COii (dry water ice); iodine (Figure \(\PageIndex{two}\)); naphthalene, a substance used to protect woolen wearable confronting moths; and ane,4-dichlorobenzene. Every bit shown in Effigy \(\PageIndex{i}\), the enthalpy of sublimation of a substance is the sum of its enthalpies of fusion and vaporization provided all values are at the aforementioned T; this is an application of Hess'south police.
\[ΔH_{sub} =ΔH_{fus} +ΔH_{vap} \label{Eq1}\]
Fusion, vaporization, and sublimation are endothermic processes; they occur only with the absorption of heat. Anyone who has ever stepped out of a pond pool on a absurd, informal twenty-four hours has felt the heat loss that accompanies the evaporation of water from the skin. Our bodies use this same phenomenon to maintain a constant temperature: we perspire continuously, fifty-fifty when at rest, losing virtually 600 mL of water daily by evaporation from the skin. Nosotros also lose about 400 mL of water every bit h2o vapor in the air we exhale, which likewise contributes to cooling. Refrigerators and air-conditioners operate on a similar principle: rut is captivated from the object or area to be cooled and used to vaporize a low-boiling-point liquid, such as ammonia or the chlorofluorocarbons (CFCs) and the hydrofluorocarbons (HCFCs). The vapor is and then transported to a different location and compressed, thus releasing and dissipating the estrus. Likewise, ice cubes efficiently cool a drink non because of their depression temperature but because heat is required to convert ice at 0°C to liquid water at 0°C.
Temperature Curves
The processes on the right side of Figure \(\PageIndex{1}\)—freezing, condensation, and deposition, which are the reverse of fusion, sublimation, and vaporization—are exothermic. Thus heat pumps that apply refrigerants are essentially air-conditioners running in opposite. Estrus from the surround is used to vaporize the refrigerant, which is then condensed to a liquid in coils within a house to provide estrus. The free energy changes that occur during phase changes can exist quantified by using a heating or cooling curve.
Heating Curves
Effigy \(\PageIndex{3}\) shows a heating bend, a plot of temperature versus heating time, for a 75 g sample of water. The sample is initially water ice at 1 atm and −23°C; every bit heat is added, the temperature of the ice increases linearly with time. The slope of the line depends on both the mass of the ice and the specific heat (C s) of water ice, which is the number of joules required to raise the temperature of one 1000 of ice by 1°C. As the temperature of the ice increases, the water molecules in the ice crystal absorb more and more free energy and vibrate more than vigorously. At the melting point, they have enough kinetic energy to overcome attractive forces and movement with respect to ane another. As more heat is added, the temperature of the system does not increase further but remains abiding at 0°C until all the ice has melted. Once all the ice has been converted to liquid water, the temperature of the water once again begins to increase. Now, all the same, the temperature increases more slowly than before because the specific rut chapters of h2o is greater than that of ice. When the temperature of the water reaches 100°C, the water begins to boil. Here, also, the temperature remains constant at 100°C until all the water has been converted to steam. At this point, the temperature again begins to rise, merely at a faster rate than seen in the other phases because the heat capacity of steam is less than that of water ice or h2o.
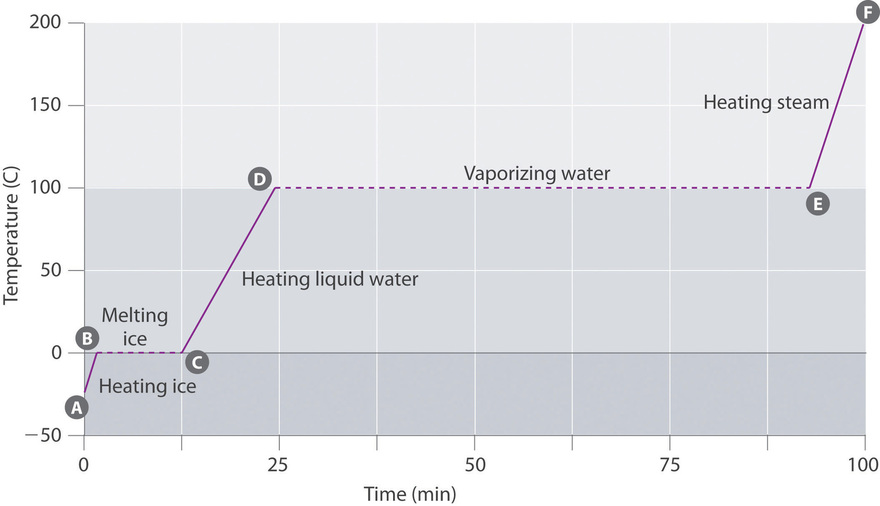
Thus the temperature of a system does not change during a phase change. In this example, every bit long equally even a tiny amount of ice is present, the temperature of the system remains at 0°C during the melting process, and as long every bit fifty-fifty a small corporeality of liquid water is present, the temperature of the arrangement remains at 100°C during the humid process. The charge per unit at which heat is added does non bear upon the temperature of the ice/water or h2o/steam mixture considering the added heat is existence used exclusively to overcome the bonny forces that hold the more condensed phase together. Many cooks retrieve that nutrient will cook faster if the estrus is turned up higher so that the water boils more rapidly. Instead, the pot of water will boil to dryness sooner, simply the temperature of the water does not depend on how vigorously information technology boils.
The temperature of a sample does not change during a phase change.
If heat is added at a constant rate, as in Effigy \(\PageIndex{3}\), then the length of the horizontal lines, which represents the time during which the temperature does non modify, is directly proportional to the magnitude of the enthalpies associated with the stage changes. In Effigy \(\PageIndex{3}\), the horizontal line at 100°C is much longer than the line at 0°C because the enthalpy of vaporization of water is several times greater than the enthalpy of fusion.
A superheated liquid is a sample of a liquid at the temperature and pressure level at which information technology should be a gas. Superheated liquids are not stable; the liquid will somewhen eddy, sometimes violently. The phenomenon of superheating causes "bumping" when a liquid is heated in the laboratory. When a exam tube containing water is heated over a Bunsen burner, for case, one portion of the liquid can easily get too hot. When the superheated liquid converts to a gas, it can push or "bump" the residuum of the liquid out of the test tube. Placing a stirring rod or a small piece of ceramic (a "boiling chip") in the test tube allows bubbles of vapor to form on the surface of the object and then the liquid boils instead of becoming superheated. Superheating is the reason a liquid heated in a smooth loving cup in a microwave oven may not boil until the loving cup is moved, when the movement of the loving cup allows bubbling to course.
Cooling Curves
The cooling curve, a plot of temperature versus cooling time, in Figure \(\PageIndex{4}\) plots temperature versus time as a 75 k sample of steam, initially at ane atm and 200°C, is cooled. Although we might await the cooling curve to be the mirror epitome of the heating curve in Figure \(\PageIndex{3}\), the cooling bend is not an identical mirror paradigm. Equally heat is removed from the steam, the temperature falls until information technology reaches 100°C. At this temperature, the steam begins to condense to liquid water. No further temperature change occurs until all the steam is converted to the liquid; then the temperature once more decreases as the h2o is cooled. We might expect to achieve another plateau at 0°C, where the water is converted to water ice; in reality, however, this does not always occur. Instead, the temperature often drops below the freezing point for some time, as shown by the little dip in the cooling curve below 0°C. This region corresponds to an unstable form of the liquid, a supercooled liquid. If the liquid is immune to stand, if cooling is continued, or if a small crystal of the solid stage is added (a seed crystal), the supercooled liquid will convert to a solid, sometimes quite suddenly. As the water freezes, the temperature increases slightly due to the estrus evolved during the freezing process then holds abiding at the melting point every bit the balance of the h2o freezes. After, the temperature of the water ice decreases again as more heat is removed from the system.
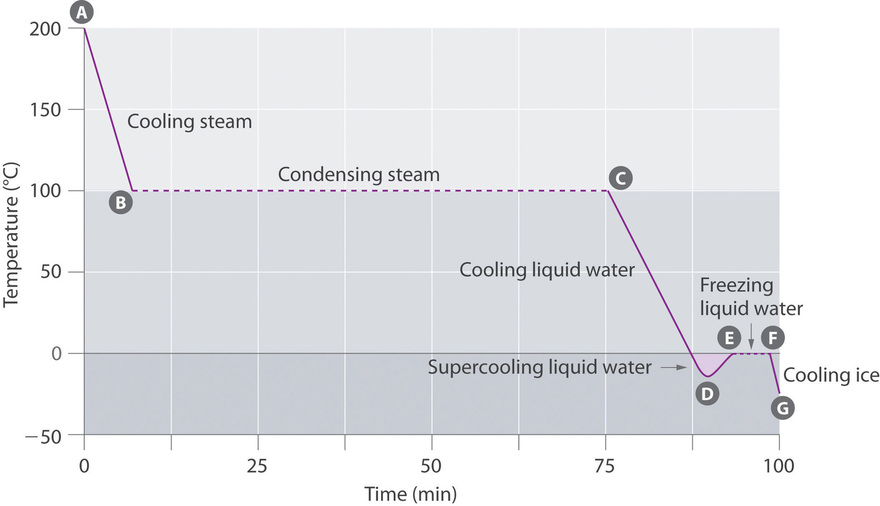
Supercooling furnishings have a huge impact on Earth's climate. For example, supercooling of water droplets in clouds can prevent the clouds from releasing precipitation over regions that are persistently barren equally a result. Clouds consist of tiny droplets of h2o, which in principle should be dumbo enough to autumn as rain. In fact, however, the droplets must amass to achieve a sure size earlier they can autumn to the ground. Usually a minor particle (a nucleus) is required for the aerosol to aggregate; the nucleus tin can be a dust particle, an ice crystal, or a particle of silver iodide dispersed in a cloud during seeding (a method of inducing rain). Unfortunately, the small aerosol of water mostly remain as a supercooled liquid downwardly to nearly −x°C, rather than freezing into ice crystals that are more suitable nuclei for raindrop germination. 1 approach to producing rainfall from an existing cloud is to cool the water droplets so that they crystallize to provide nuclei around which raindrops can grow. This is best washed past dispersing small granules of solid CO2 (dry water ice) into the cloud from an airplane. Solid CO2 sublimes directly to the gas at pressures of 1 atm or lower, and the enthalpy of sublimation is substantial (25.3 kJ/mol). As the CO2 sublimes, information technology absorbs rut from the deject, often with the desired results.
A Video Discussing the Thermodynamics of Phase Changes. Video Source: https://youtu.be/Uf2mAuP1BZY
Example \(\PageIndex{ane}\): Cooling Tea
If a 50.0 1000 ice cube at 0.0°C is added to 500 mL of tea at 20.0°C, what is the temperature of the tea when the ice cube has just melted? Assume that no heat is transferred to or from the surroundings. The density of water (and iced tea) is ane.00 g/mL over the range 0°C–20°C, the specific heats of liquid water and ice are iv.184 J/(one thousand•°C) and ii.062 J/(chiliad•°C), respectively, and the enthalpy of fusion of ice is six.01 kJ/mol.
Given: mass, volume, initial temperature, density, specific heats, and \(ΔH_{fus}\)
Asked for: concluding temperature
Strategy
Substitute the given values into the general equation relating rut gained (by the ice) to heat lost (by the tea) to obtain the concluding temperature of the mixture.
Solution
When two substances or objects at different temperatures are brought into contact, heat will flow from the warmer one to the cooler. The amount of heat that flows is given by
\[q=mC_sΔT \nonumber\]
where \(q\) is oestrus, \(one thousand\) is mass, \(C_s\) is the specific heat, and \(ΔT\) is the temperature change. Eventually, the temperatures of the two substances will go equal at a value somewhere between their initial temperatures. Calculating the temperature of iced tea subsequently adding an ice cube is slightly more complicated. The full general equation relating oestrus gained and estrus lost is notwithstanding valid, merely in this case we as well have to accept into business relationship the amount of oestrus required to melt the water ice cube from ice at 0.0°C to liquid water at 0.0°C.
The amount of heat gained past the ice cube as it melts is determined past its enthalpy of fusion in kJ/mol:
\[q=nΔH_{fus} \nonumber\]
For our l.0 m ice cube:
\[\begin{align*} q_{water ice} &= 50.0 chiliad⋅\dfrac{ane\: mol}{18.02\:g}⋅vi.01\: kJ/mol \\[4pt] &= 16.7\, kJ \end{align*}\]
Thus, when the ice cube has just melted, it has absorbed 16.7 kJ of oestrus from the tea. Nosotros can then substitute this value into the showtime equation to decide the change in temperature of the tea:
\[q_{tea} = - 16,700 J = 500 mL⋅\dfrac{1.00\: yard}{1\: mL}⋅4.184 J/(g•°C) ΔT \nonumber\]
\[ΔT = - 7.98 °C = T_f - T_i \]
\[T_f = 12.02 °C \]
This would be the temperature of the tea when the ice cube has just finished melting; nonetheless, this leaves the melted ice still at 0.0°C. We might more than practically desire to know what the concluding temperature of the mixture of tea will be once the melted ice has come up to thermal equilibrium with the tea. To determine this, we can add one more step to the calculation by plugging in to the full general equation relating heat gained and heat lost once more:
\[\begin{align*} q_{water ice} &= - q_{tea} \\[4pt] q_{ice} &= m_{water ice}C_sΔT = 50.0g⋅four.184 J/(g•°C)⋅(T_f - 0.0°C) \\[4pt] &= 209.2 J/°C⋅T_f \end{align*}\]
\[q_{tea} = m_{tea}C_sΔT = 500g⋅4.184 J/(g•°C)⋅(T_f - 12.02°C) = 2092 J/°C⋅T_f - 25,150 J \]
\[209.2 J/°C⋅T_f = - 2092 J/°C⋅T_f + 25,150 J \]
\[2301.2 J/°C⋅T_f = 25,150 J \]
\[T_f = 10.9 °C \]
The concluding temperature is in between the initial temperatures of the tea (12.02 °C) and the melted ice (0.0 °C), then this reply makes sense. In this example, the tea loses much more estrus in melting the ice than in mixing with the cold water, showing the importance of accounting for the rut of phase changes!
Exercise \(\PageIndex{1}\): Death by Freezing
Suppose y'all are overtaken by a blizzard while ski touring and you take refuge in a tent. Yous are thirsty, just you forgot to bring liquid water. Y'all have a selection of eating a few handfuls of snow (say 400 g) at −5.0°C immediately to quench your thirst or setting upwards your propane stove, melting the snow, and heating the h2o to trunk temperature before drinking information technology. You call up that the survival guide y'all leafed through at the hotel said something about non eating snowfall, just you cannot call up why—subsequently all, it's simply frozen water. To understand the guide's recommendation, calculate the amount of rut that your body volition take to supply to bring 400 g of snow at −5.0°C to your torso'due south internal temperature of 37°C. Use the data in Example \(\PageIndex{1}\)
- Answer
-
200 kJ (4.one kJ to bring the ice from −five.0°C to 0.0°C, 133.six kJ to melt the ice at 0.0°C, and 61.9 kJ to bring the water from 0.0°C to 37°C), which is free energy that would not have been expended had you lot first melted the snow.
Summary
Fusion, vaporization, and sublimation are endothermic processes, whereas freezing, condensation, and deposition are exothermic processes. Changes of country are examples of stage changes, or phase transitions. All phase changes are accompanied by changes in the energy of a system. Changes from a more-ordered state to a less-ordered state (such every bit a liquid to a gas) are endothermic. Changes from a less-ordered state to a more-ordered state (such as a liquid to a solid) are always exothermic. The conversion of a solid to a liquid is called fusion (or melting). The energy required to melt ane mol of a substance is its enthalpy of fusion (ΔH fus). The free energy modify required to vaporize 1 mol of a substance is the enthalpy of vaporization (ΔH vap). The direct conversion of a solid to a gas is sublimation. The corporeality of energy needed to sublime 1 mol of a substance is its enthalpy of sublimation (ΔH sub) and is the sum of the enthalpies of fusion and vaporization. Plots of the temperature of a substance versus heat added or versus heating fourth dimension at a constant rate of heating are chosen heating curves. Heating curves relate temperature changes to phase transitions. A superheated liquid, a liquid at a temperature and pressure at which it should be a gas, is non stable. A cooling curve is not exactly the opposite of the heating bend because many liquids do not freeze at the expected temperature. Instead, they course a supercooled liquid, a metastable liquid phase that exists below the normal melting bespeak. Supercooled liquids ordinarily crystallize on standing, or adding a seed crystal of the same or another substance can induce crystallization.
seidmandishents74.blogspot.com
Source: https://chem.libretexts.org/Courses/Bellarmine_University/BU:_Chem_104_%28Christianson%29/Phase_1:_The_Phases_of_Matter/3:_Phase_Changes/3.2:_Energy_of_Phase_Changes
0 Response to "What is the source of energy that changes water from liquid to gas"
Post a Comment